Author: Maryland Franklin, PhD, Vice President, Scientific Development
Date: February 2016
Preclinically, the murine CT26 colon carcinoma line has become a platform model for evaluating the potential of drug combinations with immune checkpoint inhibitor antibodies. It is a highly immunogenic tumor and tends to show objective response rates to a number of commercially available checkpoint inhibitors. At Labcorp, we have run this model more than a dozen times in the past nine months, with additional studies being scheduled regularly.
An estimated one in 20 people will develop colon cancer, making it one of the most frequently diagnosed cancers both in men and women. Due to improved screening methods, the overall incidence of colon cancer has declined during the past 20 years. However, it is still projected that only 40% are diagnosed with early stage disease and that 50% of all patients will also develop metastatic disease. Thus, colon cancer remains an important and active area of oncology drug discovery and development. Surgery is the most common treatment option for patients; however, many also receive chemotherapy and sometimes chemotherapy in combination with radiation therapy. Unless diagnosed early, five-year survival rates are still quite low. Today a number of clinical trials are ongoing to evaluate the potential of immunotherapies in colon cancer.
Preclinically, the murine CT26 colon carcinoma line has become a platform model for evaluating the potential of drug combinations with immune checkpoint inhibitor antibodies. It is a highly immunogenic tumor and tends to show objective response rates to a number of commercially available checkpoint inhibitors. We have run this model more than a dozen times in the past nine months, with additional studies being scheduled regularly.
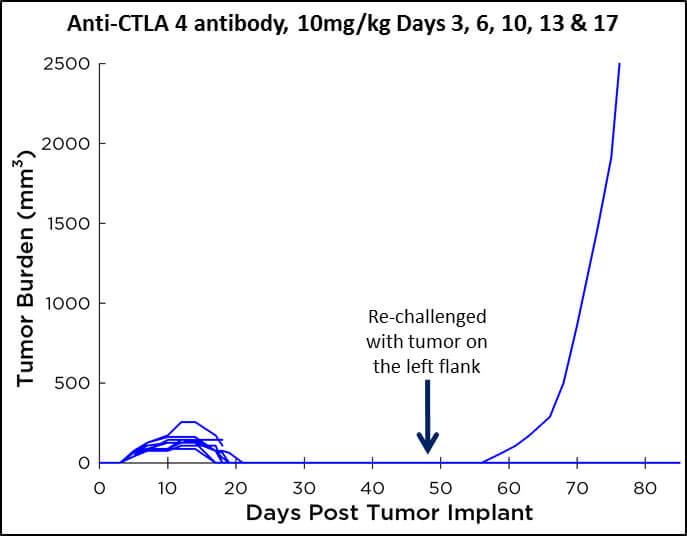
CT26 Responsive to anti-CTLA4 Antibody Treatment
The CT26 model is highly responsive to anti-CTLA4 antibody treatment with 100% of treated mice showing tumor regressions. In studies where we evaluate long term survival we find that about 70-90% of the treated mice will develop long term cures and that re-implantation of CT26 demonstrates primarily lack of tumor re-growth, suggesting the development of a memory immune response.
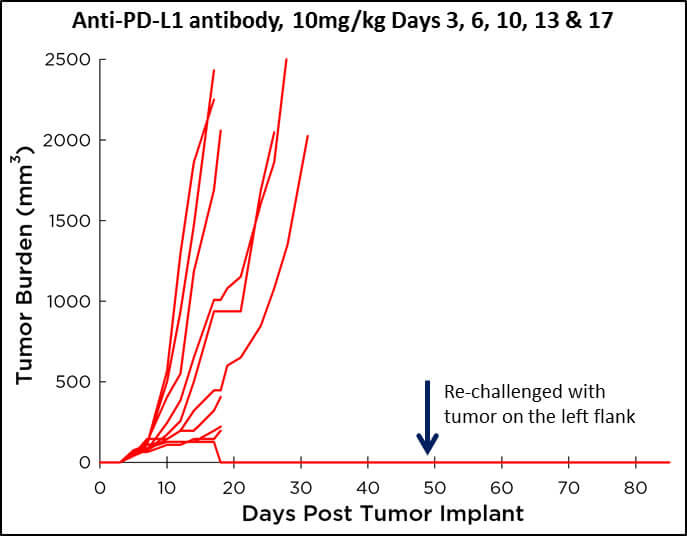
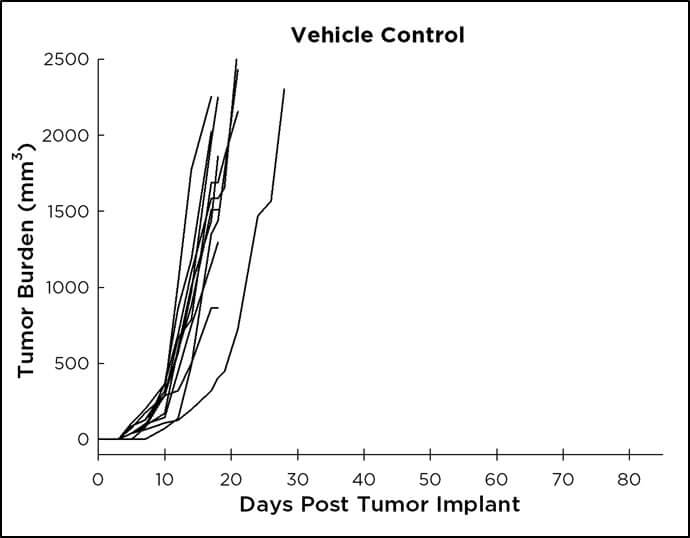
CT26 and anti-PD-L1 Treatment
In contrast to treatment with anti-CTLA4 antibody, when we treat CT26 tumor bearing mice with anti-PD-L1 antibody we find that only a small percentage of mice will respond to treatment. A few mice are capable of completely eradicating the tumor following treatment with anti-PD-L1 antibody; and when re-challenged, these mice are cured and no tumor regrowth is observed. In most cases though, we find that 40% of mice demonstrate a period of stable disease, followed by tumor outgrowth that is delayed vs. vehicle or isotype control treated mice. Interestingly, we find a proportion of mice that are completely non-responsive to anti-PD-L1 treatment, much like the clinical setting. This model, with anti-PD-L1 treatment, has proven useful in many drug combination studies.
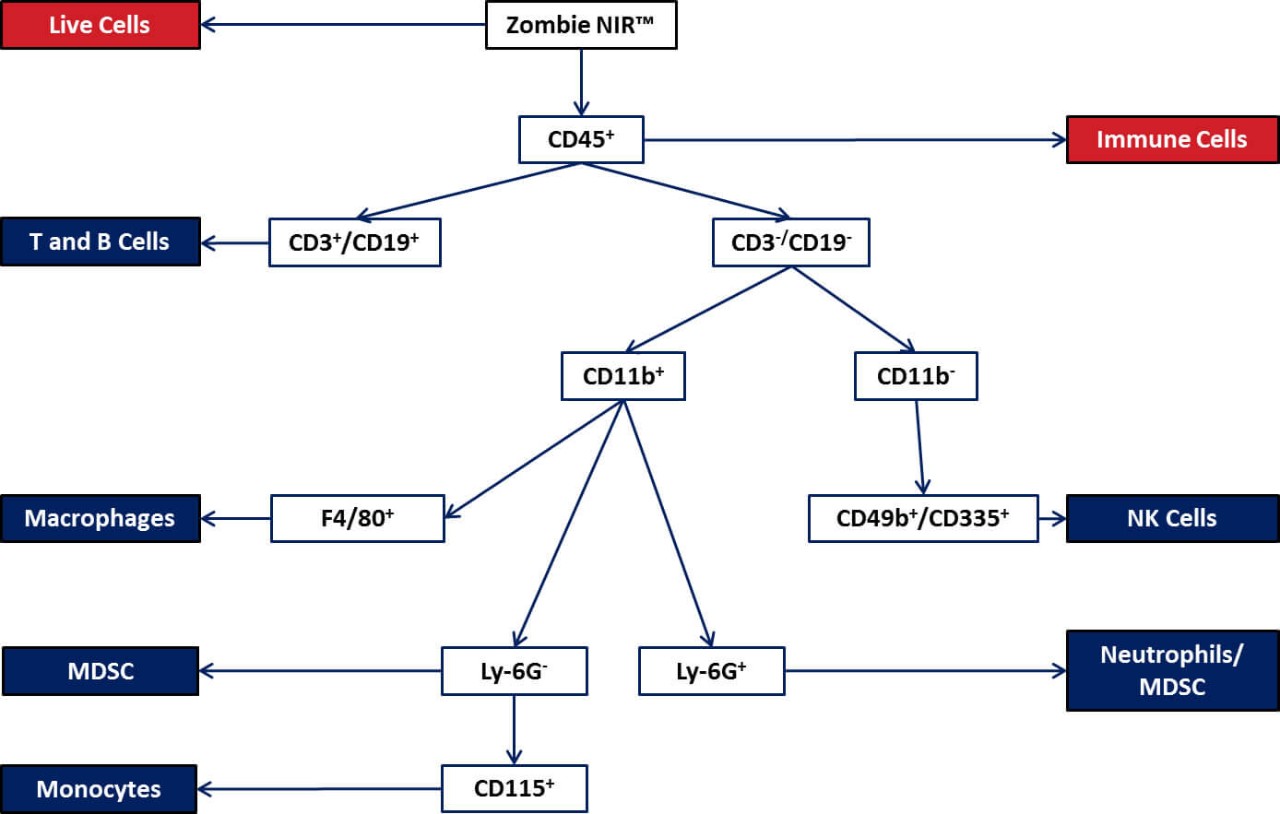
CT26 and Flow Cytometry
In addition to evaluating efficacy of anti-cancer drug combinations this model has proven useful in the assessment of phenotypic changes in the immune system. To this end we have evaluated CT26 tumors for a number of fundamental tumor infiltrating leukocytes with our Attune NxT® flow cytometer.
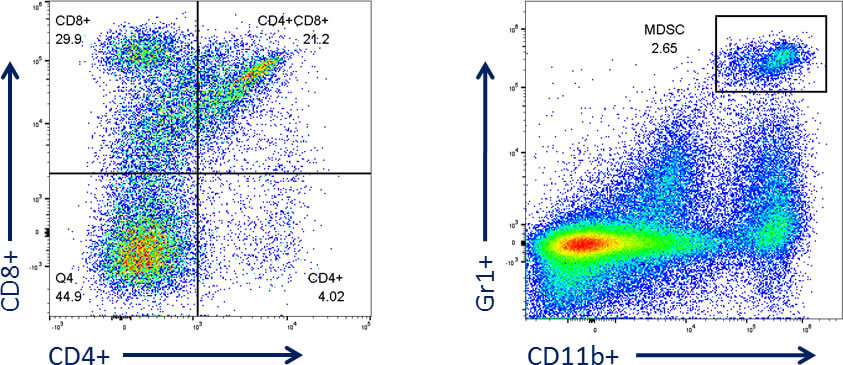
Additionally, to this basic analysis, we have also established standard panels for a more complex variety of cell populations.
Contact us today for more information on our CT26 model, other syngeneic tumor models, and our complex immuno-phenotyping panels.
References
Connect
Let's start a conversation
Contact Us